News
A new standard to help with the fight against blindness
Glaucoma is the leading cause of blindness, and now an EMPIR project has provided metrological validation for instruments that detect the main risk factor
The World Health Organization has named glaucoma as the leading cause of irreversible vision loss, responsible for 37.5 % of all blind people world-wide.
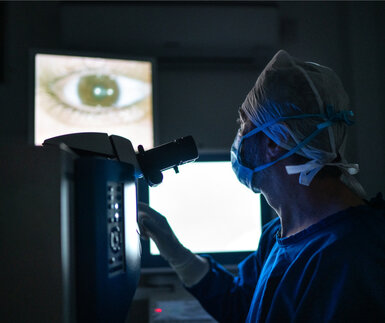
In its early stages glaucoma has no symptoms and can only be detected by an increase in intraocular pressure (IOP), the pressure inside the eye caused by fluid build-up.
As IOP is the only treatable risk factor for the disease it is essential that medical instruments that measure IOP are metrologically validated.
However, this isn’t the case for IOP non-contact measuring devices called tonometers. Despite being fast and easy to use – with eye pressure measured by ‘gusts’ of air blown at the eye – there is no universal standard for calibrating these devices. Each manufacturer uses in-house standards for calibration making measurement comparison between different types of instrument challenging. There is also a lack of critical metrological parameters such as repeatability, stability and measurement uncertainty.
This lack of independent metrological calibration can potentially result in imprecise measurements affecting the diagnosis of the disease and the subsequent treatment of glaucoma.
Thanks to the EMPIR project Developing research capabilities for traceable intraocular pressure measurements (16RPT03, InTense) the situation is now changing.
During the now completed project the Slovak University of Technology in Bratislava (STU BA) developed a virtual digital model of the eye’s cornea which formed the basis for the construction of a real mechanical model.
This ‘artificial eye’ – consisting of a piston pressure generator and a pressure chamber with an artificial cornea – was built to act as a new transfer standard for tonometer calibration.
The calibration ability of the new standard was tested in an interlaboratory comparison against a set of silicone eyes commonly used in calibration and verification procedures, by STU BA and CMI, the Czech National Metrology Institute (NMI) and supported by SMU, the NMI of Slovakia. After the comparison a clinical trial took place, at the Ružinov Hospital in Bratislava, Slovakia.
These comparisons confirmed the high potential of the eye model, setting the stage for the successful development of the artificial eye as a universal transfer standard in the near future.
The new standard, along with the new calibration facility for calibration of eye tonometers also developed in the project, will go a long way to providing better protection against glaucoma in Europe.
Dominik Pražák (CMI) who coordinated the highly successful project has said about the work :
” We hope that this universal transfer-standard will one day replace the fragmented, specialized solutions and will contribute to a uniform, easier and cheaper assurance of the metrological traceability of the intraocular pressure measurements.”
This EMPIR project is co-funded by the European Union's Horizon 2020 research and innovation programme and the EMPIR Participating States.
Want to hear more about EURAMET?
Information
- EMPIR,
- Research Potential,
- EMN Traceability in Laboratory Medicine,
Developing a metrologically-based field assessment of glare and obtrusive light more
Standardising industrial procedures for the magnetic properties of devices leading to the improved quality of a wide variety of products more
Implementing quantum-based pressure measurement techniques in European industries more
Developing reference materials for mass spectroscopy to monitor radioactive and stable isotope pollution in the environment more
Development of the metrological network needed to realise and implement 6G technology more