Traceability in Laboratory Medicine: supporting the COVID-19 response

The coronavirus pandemic has stopped large parts of the world in its tracks. Presently, countries across the globe are experiencing different stages of the wave of the infection, leading to varying levels of lockdown reinstallments and attempts to resume normality.
Importantly, governmental decisions to ease lockdown restrictions are strongly influenced by the rate of accrual of new infections for any given population.
Reliable testing results are thus essential to delineate true cases from other non-coronavirus ‘flu-like’ symptoms - even at the very onset of the infection and for those with just mild symptoms.
In response to an urgent global need, healthcare professionals, laboratory medicine specialists and diagnostic assay developers worldwide have sprung into action - and today a multitude of COVID-19 tests continue to be developed, including those that cater specially for the European market.
Members of EURAMET's Metrology Network for Traceability in Laboratory Medicine (EMN TraceLabMed) are key drivers behind these efforts. The network is proud to assist on several levels:
- providing metrological expertise;
- coordinating European proficiency testing schemes;
- providing reference values for common reference materials; and much more.
Traceability for COVID-19 tests
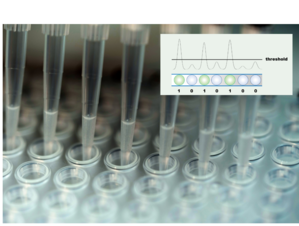
At the onset of the COVID-19 infection, the virus multiplies in its human host, shedding multiple copies of its heritable material (its RNA) throughout the system of the host. This RNA can be extracted from a patient's sample together with massive amounts of other nucleic acids from its human host. Highly specific nucleic acid amplification techniques (polymerase chain reaction, PCR) are needed to find the rare traces of the virus. It is the presence of viral RNA traces in the PCR that then confirms the infection.
Over the course of the infection, antibodies against viral surface proteins will form in the patient and can be identified even if the acute phase of the infection has passed. The testing procedure is therefore not as straightforward as often assumed. Given many of these tests are developed ad hoc and might not be particularly sensitive in patients, it is not surprising that many countries have resorted to testing serious cases of the infection only.
This lack of sensitivity in testing can result in an inevitable miss of a large fraction of the infections at a given time point. Results can also vary wildly for different test providers, even if material from the same patient is repeatedly analysed with different tests. Given the need for quick test results, many tests have been developed and have gained European market access under the still-applicable in vitro diagnostic directive (IVDD).
Comparing test quality

The quality of COVID-19 viral and antibody diagnostic tests can be compared through the use of well-characterized common reference materials. Important questions concerning the quality of testing that provide insight into the most preferable tests overall - including the limits of detection and quantification and the precision of measurements of known reference substance amounts in a sample - are all typically answered using metrological tools.
TraceLabMed members play a significant role here, and their work on enhancing the quality and validity of test diagnoses will support healthcare systems and contribute to the worldwide coronavirus response.
News Stories
NIBSC develops new reference materials to support the pandemic response
The UK’s National Institute for Biological Standards and Control (NIBSC) has been a critical source of research support during our global pandemic. The institute is an important member of EURAMET’s metrology network for traceability in laboratory medicine (EMN TraceLabMed), and has recently developed new reference materials and reagents that will underpin the accurate diagnoses, development of vaccines and other medicines for COVID-19. Read more >>
Laboratory Medicine EMN strengthens relationships with key medical organisations
Laboratory medicine results, such as those dervied from regularly performed in vitro diagnostic (IVD) tests, affect medical decisions that are made throughout a patient's lifetime. To ensure tests are able to provide reliable results for aiding clinical diagnoses, it is essential for medical laboratories to demonstrate measurement traceability.
Since its launch, EURAMET's TraceLabMed network has been working to engage with internationally recognised stakeholders in laboratory medicine, to improve communciation within this entire subsect of the medical community, and ultimately, to strengthen the fundamental property of measurement traceability for diagnostic tests. Read more >>
Members of Traceability in Laboratory Medicine network support COVID-19 response
EURAMET’s TraceLabMed members have been making sure to play their part in the global response to our coronavirus pandemic. From the development of metrological tools to support better traceability and reliability for COVID-19 tests, to the participation of network members in international ring trials for infection diagnostic testing; TraceLabMed continues to provide essential support to the rest of the laboratory medicine community, as well as healthcare professionals around the world. Read more >>
The NIBSC, a member of TraceLabMed, supports vital COVID-19 research
An influential member of EURAMET’s network for traceability in laboratory medicine - the UK’s National Institute for Biological Standards and Control (NIBSC) - has helped to fast-track the development of key reference materials and research reagents for COVID-19. The institute’s work with biological standards, calibrant materials for the WHO and their specialised centre for AIDS reagents will all support the development of treatments and vaccines for the coronavirus pandemic. Read more >>
TraceLabMed and EMPIR projects make vital contributions to COVID-19 testing
European metrology institutes from EURAMET’s TraceLabMed network are providing essential support for both viral and antibody testing for COVID-19. EURAMET projects like ‘AntiMicroResist’ and ‘Bio-stand’ have brought together the collaborative power of metrology researchers to develop inter-laboratory comparison methods and new standards to support the implementation and evaluation of COVID-19 testing techniques. TraceLabMed members have also joined forces in inter-laboratory studies for measuring the SARS-CoV-2 genome and for validating antibody tests. Read more >>

