Metrology and innovation for early diagnosis and accurate stratification of patients with neurodegenerative diseases
Improved accuracy for early-stage neurodegenerative disease diagnostic tools
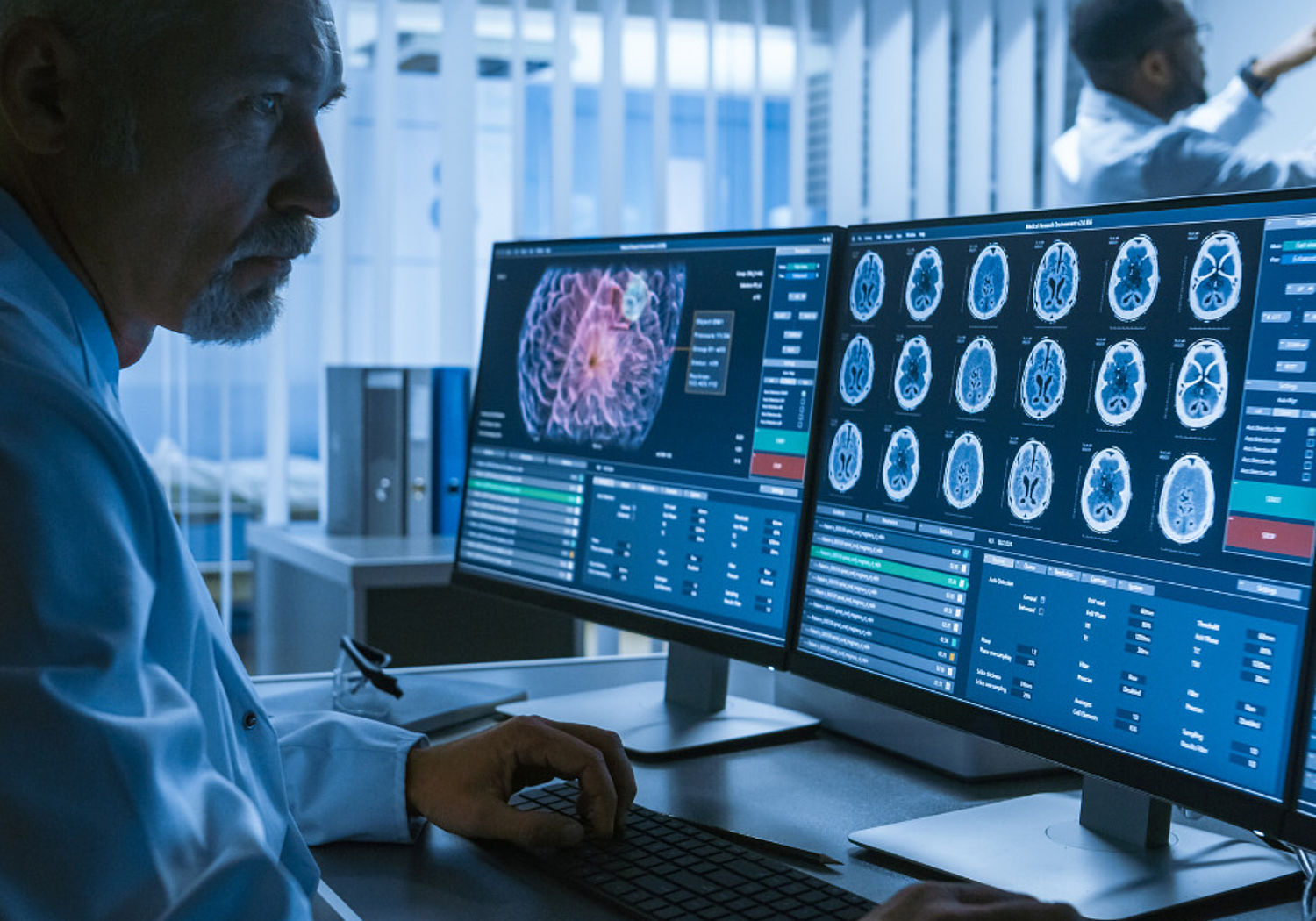
Neurodegenerative diseases (NDDs) are a major health challenge for society; over 9.9 million new cases of dementia are identified worldwide each year, while annual care costs in Europe currently run to €130 billion. More accurate and earlier diagnosis would enable more effective treatment and improve forecasting of disease progression. Improved evaluation of NDD therapies is also needed before clinical uptake can occur.
This project aimed to develop the most promising, minimally invasive, measurement methods for early NDD diagnosis and supported development of targeted (patient-specific) therapies. The project will lead to improvements in the resolution of magnetic resonance imaging (MRI) and other scanning technologies, validate biomarker measurements and develop a smartphone based cognitive test app – all designed for eventual clinical use. Measurement protocols will support standardisation for early NDD diagnosis and new therapies, enabling development of screening programmes for improved quality of life.
This project builds on from EMPIR project 15HLT04 NeuroMet.
Scientific Reports
Clinical Chemistry and Laboratory Medicine (CCLM)
Measurement: Sensors
Magnetic Resonance in Medicine
International Journal of Molecular Sciences
Measurement: Sensors
Entropy
Participating EURAMET NMIs and DIs
LGC (United Kingdom)
LNE (France)
PTB (Germany)
RISE (Sweden)
Other Participants
Centre Hospitalier Universitaire Montpellier (France)
Charite - Universitaetsmedizin Berlin (Germany)
Högskolan Kristianstad (Sweden)
Modus Outcomes SAS (France)
Stichting VUmc (Netherlands)
Universitätsmedizin Greifswald - Körperschaft des öffentlichen Rechts (Germany)
Information
